“It’s genetic. There’s nothing that can be done about it.” The idea that we have no control over our genes is a common one. In a sense, it is correct – we have no say in what genes we happen to inherit from our parents, that is beyond our control. However, we do have control over whether those genes affect our lives negatively or positively. Today, we’re going to learn how omega-3 can affect genes.
Table of Contents
What’s DNA?
The field of science known as epigenetics studies the chemical processes that turn genes on and off. As research in the field continues, it’s becoming clear that there is a lot more to our genetic attributes than simply what is present in our DNA code.
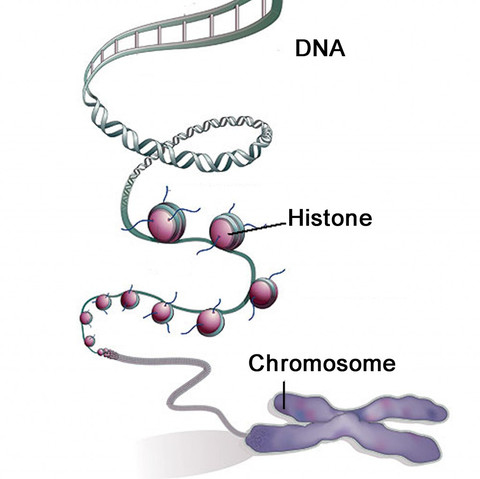
DNA is in every cell of our bodies. It is the handbook that tells the cell what to be and how to function. It is so packed with information that the only way it can fit inside a cell is by being stored in a chain and then wrapped around histones.
Histones are round proteins that are similar to a spool in appearance, and the DNA wraps around them. When the DNA is loosely wrapped and there’s space between the histones, that portion of the DNA can interact with other parts of the cell and is “turned on”. These genes that are loosely wrapped will be expressed. On the other hand, if the DNA is wrapped tightly around the histones and they are pulled together, that section of the DNA is blocked from interacting and the genes can not be expressed. It is ‘turned off’.
Genetic expression
The process of the DNA tightening or loosening around the histones is controlled by two chemical tags: methyl and acetyl.
Acetyl attaches to the histones and weakens the interaction with the DNA, causing the chain to loosen and the genes to turn on.
Methyl attaches to the DNA and strengthens the interaction, causing the DNA to wrap tighter and pull the histones together. This is called DNA methylation and it’s what turns the genes off.
The study of epigenetics seeks to find how to turn “bad” genes off and turn “good” genes on. Our DNA cannot be changed because we can neither remove nor add genes, but the expression of those genes can be influenced. Gene expression is not solely influenced by internal factors. It may also be strongly influenced by external factors such as environment, experiences, and diet. That means that regardless of what genes we inherited, the way we live and how we eat can determine how our genes act.
Omega-3 in gene expression
Diet can play a significant role in modifying the expressions of genes. Omega-3 fatty acids are already dietary recommended due to the wealth of health benefits it offers. While it is known most popularly as a heart-healthy supplement, there is already quite a bit of research showing that it also plays a role in gene expression.
In fact, the cardiovascular benefits of omega-3 may be linked to its effect on our genes. For example, one study found that omega-3 supplementation stopped the expression of cyclooxygenase-2, which is a gene that contributes to the buildup of plaque in the arteries.
Omega-3 affects epigenetics and gene expression mainly through promoting DNA methylation. The strongest benefits have been shown when omega-3 was supplemented during prenatal development.
In a 2011 study, DHA (type of omega 3) deficiency during pregnancy was linked to lower methylation in the offspring. The results of the study showed that DHA is essential for ensuring the proper amount of methylation.
This was supported by a similar study in 2013, which concentrated on women who smoked tobacco throughout their pregnancy. Nicotine and the toxins within tobacco products hinder DNA methylation, but the study showed that this complication was significantly reduced in the children of the women that had been given omega-3.
Lower methylation may result in a number of disorders because genes that were meant to be “turned off” did not react with enough methyl for the DNA to coil tightly and avoid interaction or signalling. However, there are indications that this problem can be corrected.
In the 2011 study, DHA was added into the diet and the rate of methylation returned to healthy levels. So, even if there was an omega-3 deficiency, supplementation in infancy and early childhood may reverse the lower methylation rates and regulate the gene expressions.
Omega-3, genes, and cancer
While omega-3 is important during prenatal development, its role in genetic expression continues into adulthood. One the most promising findings has been the role it can play in treating certain types of cancers. Two of the cancer types studied are ones that are often considered genetic and tend to run in families: breast cancer and prostate cancer.
When these diseases run in families, it means that certain genes with a tendency to promote malignant cell mutation are passed down. With cancer, these genes are called oncogenes. There is no way to remove these genes from the DNA, but there is a chance that they can be silenced.
In the study focusing on breast cancer, there was a notable decrease in histone activity within the cancer cells once omega-3 was administered. The study regarding prostate cancer yielded very similar results and showed a recession of the cancer. These indicate that methylation occurred and the histones became so tightly wrapped in the DNA that they were no longer able to send or receive signals. In essence, the genes were shut off and the cell stopped getting the signals to replicate or grow.
By having the genes shut off, it can lead to the death of the cancerous cell because the genes will no longer be telling it what to do or how to function. The conclusion of these studies suggested that omega-3’s methylation promoting qualities make it useful for preventing cancer, and possibly for helping to treat it as well.
Conclusion
It’s true that we are stuck with the genes we inherited, both the good and the bad, but that does not mean we are powerless in keeping them from negatively affecting our health. The field of epigenetics is still relatively new, and research into figuring out how external factors affect gene expression is still ongoing. However, there is already a significant amount of evidence that diet plays a large role and that a high-quality omega-3 supplement can have a beneficial effect on the regulation of gene expression.
💬 Something on your mind? Share your thoughts in the comments. We love hearing from curious minds.
📩 And while you’re here, join our newsletter for more smart stuff (and secret perks)!
References:
- Berquin, I., Min, Y., Wu, R., Wu, J., Perry, D., Cline, J., . . . Chen, Y. (2007). Modulation of prostate cancer genetic risk by omega-3 and omega-6 fatty acids. Journal of Clinical Investigation, 117(7), 1866-1875. Retrieved August 19, 2015, from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1890998/
- Caterina, R., & Massaro, M. (2005). Omega-3 Fatty Acids and the Regulation of Expression of Endothelial Pro-Atherogenic and Pro-Inflammatory Genes. J Membrane Biol Journal of Membrane Biology, 206(2), 103-116. Retrieved August 19, 2015, from http://link.springer.com/article/10.1007/s00232-005-0783-2
- Dimri, M., Bommi, P., Sahasrabuddhe, A., Khandekar, J., & Dimri, G. (2009). Dietary omega-3 polyunsaturated fatty acids suppress expression of EZH2 in breast cancer cells. Carcinogenesis, 31(3), 489-495. Retrieved August 19, 2015, from http://carcin.oxfordjournals.org/content/31/3/489.short
- Enzymlogic (Photographer). (2013 September 12). DNMT1 [digital image]. Retrieved from www.enzymlogic.com
- Kulkarni, A., Dangat, K., Kale, A., Sable, P., Chavan-Gautam, P., & Joshi, S. (2011). Effects of Altered Maternal Folic Acid, Vitamin B12 and Docosahexaenoic Acid on Placental Global DNA Methylation Patterns in Wistar Rats. PLoS ONE. Retrieved August 19, 2015, from http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0017706
- Lee, H., Barraza-Villarreal, A., Hernandez-Vargas, H., Sly, P., Biessy, C., Ramakrishnan, U., . . . Herceg, Z. (2013). Modulation of DNA methylation states and infant immune system by dietary supplementation with -3 PUFA during pregnancy in an intervention study. American Journal of Clinical Nutrition, 98(2), 480-487. Retrieved August 19, 2015, from http://ajcn.nutrition.org/content/98/2/480.short
- Muskiet, F., & Kemperman, R. (2006). Folate and long-chain polyunsaturated fatty acids in psychiatric disease. The Journal of Nutritional Biochemistry, 17(11), 717-727. Retrieved August 19, 2015, from http://www.jnutbio.com/article/S0955-2863(06)00027-1/abstract
- Rettner, R. (2013, June 24). Epigenetics: Definition & Examples. Retrieved August 19, 2015.
- Sable, P., Dangat, K., Joshi, A., & Joshi, S. (2012). Maternal omega 3 fatty acid supplementation during pregnancy to a micronutrient-imbalanced diet protects postnatal reduction of brain neurotrophins in the rat offspring. Neuroscience, 217, 46-55. Retrieved August 19, 2015, from http://www.sciencedirect.com/science/article/pii/S0306452212004332